Research
IRON SUPPLEMENTATION IN YOUNG IRON-DEFICIENT FEMALES CAUSES GASTROINTESTINAL REDOX IMBALANCE: PROTECTIVE EFFECT OF A FERMENTED NUTRACEUTICAL.
FILE2014
Category StomachHealthy individualIron deficiencyClinical research
IRON SUPPLEMENTATION IN YOUNG IRON-DEFICIENT FEMALES CAUSES GASTROINTESTINAL REDOX IMBALANCE: PROTECTIVE EFFECT OF A FERMENTED NUTRACEUTICAL
J Biol Regul Homeost Agents. 28(1):53-63,2014
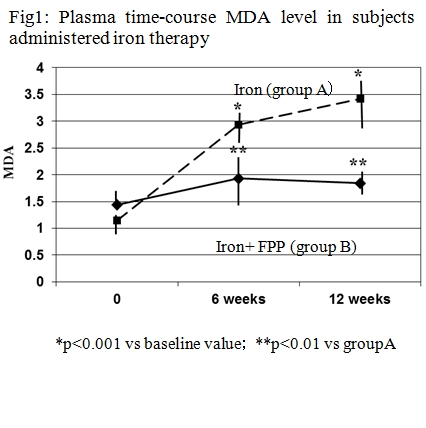
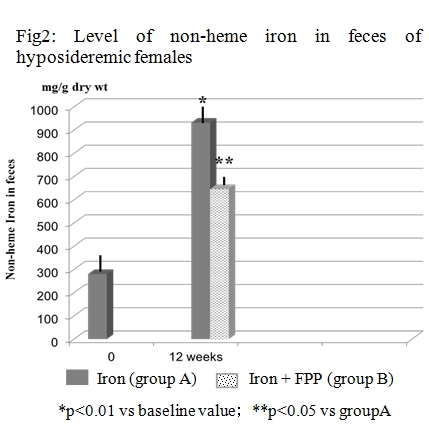
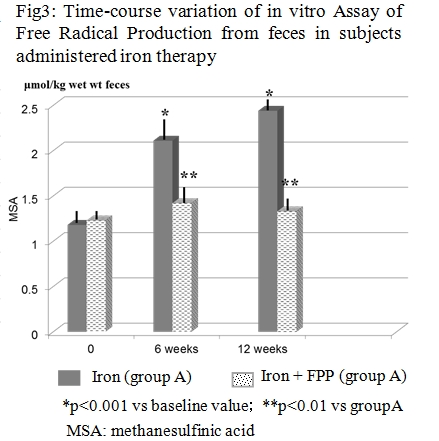
The aim of this study was to assess whether the concomitant supplementation of certified fermented papaya preparation (FPP, ORI, Gifu, Japan) together with iron supplementation could beneficially affect lipid peroxidation either systemically and at a intraluminal gut level in women with low iron stores. Treatment compliance and iron absorption was assessed as well. Fifty-two non-pregnant, fertile, non-smokers, healthy women with iron deficiency were recruited. The women were given iron supplements (100 mg Fe/d as ferrous sulfate) to be taken daily for 12 weeks (group A). Group B patients were supplemented also with 6 g/ day of a FPP. A detailed life style questionnaire was administered to all subject. Iron, ferritin, transferrin receptors (Tf R) and malondialdehyde (MDA) in plasma were measured. The RBCs lysate was used for the estimation of superoxide dismutase(SOD) and glutathione peroxidase (GPx). The total and free iron concentration as well as analysis of oxidative stress in the feces was measured. FPP-supplemented subjects showed a significantly lower degree of gastrointestinal discomforts (p< 0.05) and abolished the iron supplementation-induced increase of MDA (p< 0.001) and depletion of SOD and GPx (p< 0.01). Moreover, the nutraceutical co-administration brought about a significant reduction of gut oxidative damage and lower fecal content of total and free iron (p< 0.05 vs group A). Overall, group B showed a better TfR/ferritin ratio response (p< 0.05 vs group A). While iron supplementation maintains its clinical relevance considering that prevalence of iron deficiency among females, a careful clinical evaluation and a protective nutraceutical co-administration, as our data suggest with FPP, should be considered.
List of the related papers
Year
Name of Papers
2014
